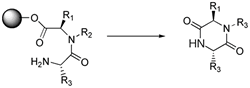Glossary of Terms Used in Combinatorial
Chemistry
[A-B]
[C] [D] [E-G]
[H-K] [L]
[M-N] [O-P] [Q] [R]
[S] [T] [U-Z]
C
Capacity: The amount of material which
may be attached to a support. May be greater than loading
due to, for example, steric effects at the solid surface.
Chemset: A collection of two or more library
members, building
blocks, or reagents; prefered notation in the Journal of
Combinatorial Chemistry14
and convenient for describing synthetic procedures on pools of compounds.
Thus 'chemset 3{1-3} denotes three members of the library produced
by the reaction of reagents 2{1-3} with starting material 1'.
Cleavage: Process of releasing compound
from solid support, thereby
permitting assay or analysis of the compound by solution-phase methods.
Dissolution of the compound following cleavage, rather than the cleavage
step itself, may be rate-limiting.
Cluster: Group of compounds which are
related by structural or behavioral properties. Organizing a set of
compounds into clusters is often used in assessing the diversity
of those compounds, or in developing SAR models. See also principal
components analysis, binning
and recursive partitioning 15-17.
Combichem: see Combinatorial
Chemistry
Combinatorial: " 1. of, relating
to, or involving combinations; 2. of, or relating to the arrangement
of, operation on, and selection of discrete elements belonging to finite
sets." (Webster's Collegiate Dictionary)
Combinatorial Chemistry:
using a combinatorial process
to prepare sets of compounds from sets of building
blocks.
Combinatorial Library: A
set of compounds prepared by combinatorial
chemistry. May consist of a collection of pools,
or sub-libraries. Its composition
may be described by the chemset
notation.
Controlled Release: see Partial
Release
Crosslinking:
Property of a solid support
prepared from polymeric materials with interconnected strands. Often
results from the inclusion of multi-functional monomers in the polymerization
reaction, e.g. divinylbenzene in polystyrene production. In such cases
the degree of crosslinking is often quoted as the proportion of the
multifunctional monomer in the reaction mixture. The extent of crosslinking
is important for physical properties of the solid support, such as the
propensity to swell in different solvents 18.
Cyclative Cleavage: Cleavage
resulting from intramolecular reaction at the linker which results in
a cyclized product. The cleavage may also act as a purification if resin-bound
side-products are incapable of cyclizing, and thus remain attached to
the solid support on release
of the desired material 9.
Diketopiperazine formation, as shown below, is one well-known example
of cyclative cleavage 19,20.