Glossary of Terms Used in Combinatorial
Chemistry
[A-B]
[C] [D] [E-G]
[H-K] [L]
[M-N] [O-P] [Q] [R]
[S] [T] [U-Z]
O-P
Omission Library: Strategy for
identifying active library members by the systematic omission of building
blocks from mixtures. Observation of reduced activity in a certain pool
suggests that the building block which was omitted in that pool contributes
to activity 66.
Orthogonality: (a) Property of
protecting groups or linkers allowing
removal, modification, or cleavage
of one such without affecting others; (b) pooling
strategy whereby library members
are incorporated in more than one pool, and are mixed with a different
set of other members in each pool. Thus a hit
results in two or more active pools with only one member in common 67,68.
Parallel Synthesis: Strategy
whereby sets of discrete compounds are prepared simultaneously in arrays
of physically separate reaction vessels or microcompartments without
interchange of intermediates during the assembly process. Contrast Pool/Split.
Partial Library: Partly assembled
library, or portion thereof which is reserved to be completed once initial
property relationships have been identified. For instance, part of an
intermediate pool may not be treated
with the final building block
until the optimal residue at the
final position is known, thus avoiding the need to prepare that pool
from the starting materials 69.
Partial Release: Cleavage
process designed to release compound from solid
support in discrete portions e.g. by using orthogonal
linkers or by controlled application of cleavage reagent or
condition. Photocleavable linkers, such as that shown below, are a particularly
convenient application since cleavage may be controlled by simply turning
on or off a lamp 70,71.
Also controlled release,
tiered release.
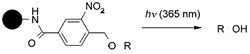
PEG: see Poly(ethylene
glycol)
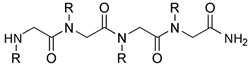
Peptoid: Oligomer consisting of repeating
N-substituted glycine units 72,73.
Phage Display: Use of genetically
engineered phage to present peptides as segments of their native surface
proteins. Peptide libraries may be produced by populations of phage
with different gene sequences 74-76.
Pharmacophore: 'The ensemble of
steric and electronic factors which are necessary to insure supramolecular
interactions with a specific biological target structure' 77-78.
Phase Switch: Strategy for compound
isolation whereby the desired material is rendered sufficiently different
from reagents, side products, and other impurities that it may be separated
from them by simple physical processes such as filtration or extraction.
May be achieved by the attachment of a tag,
such as a highly fluorinated component (see fluorous
synthesis), or other sequestration-enabling
reagent 43.
Photolithography: Process by
which selective masking generates light patterns
which direct chemical transformations to certain areas of a photosensitive
surface. Coupling of different building blocks to discrete sites may
give rise to spatially addressable
arrays of compounds 61.
Pin: An elongated device in which the tip
acts as a solid support. An array of pins may typically be held such
that sets of pins may be simultaneously inserted or retracted from solvents
or reagents allowing library preparation by parallel
synthesis 8.
Point of Diversity: Portion of a molecule,
or step in a synthetic scheme, where different building
blocks may be introduced.
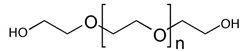
Poly(ethylene glycol) (PEG):
Polymer which has been applied both as soluble support and (as
a graft copolymer with a polystyrene matrix) as a linker
for combinatorial synthesis. See TentaGel,
ArgoGel. The soluble support may
have hydroxyls at both termini, or one or both may be capped or modified
with additional functionality 59.
Pool: (a) A sub-library;
(b) process of combining and mixing library components or sub-libraries.
See pool/split.
Pool/Split: (split/pool;
split&mix;
divide, couple, recombine;
portion/mix) Strategy for assembly of a combinatorial
library. The solid support is divided into portions,
each of which is subjected to reaction with a single building
block. Pooling of these portions results in a single batch of
solid support bearing a mixture of components. Repetition of the divide,
couple, recombine processes results in a library where each discrete
particle of solid support carries a single library member, and the number
of members is equal to the product of the number of building blocks
incorporated at each step (i.e. fully
combinatorial) 79,80.
Positional Scan: Strategy for
identifying individual compounds of interest from a library whereby
a collection of sub-libraries
is prepared, equal in number to the total number of
building blocks used in the entire library. In each pool
one point of diversity is held constant
by incorporating a single building block while the other positions use
all possible building blocks 81.
Preformed Scaffold: A scaffold
which is incorporated into the library as a unit. Compare in
situ scaffold.
Principal Components Analysis:
Computational approach to reducing the complexity of, for example, a
set of descriptors, by identifying
those features which provide the major contributions to observed properties,
and thus reducing the dimensionality of the relevant property
space 30,82.
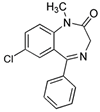
Privileged Structure: Substructural
feature which confers desirable (often drug-like) properties on compounds
containing that feature. Often consists of a semi-rigid scaffold
which is able to present multiple hydrophobic residues
without undergoing hydrophobic collapse, e.g. diazepam (below) in
which the diphenylmethane moiety prevents association of the aromatic
rings 83.
Property Space: Multidimensional
representation of a set of compounds in which the axes represent quantifiable
properties such as molecular weight, c LogP, molar refractivity,
etc., and individual compounds are represented by a vector or set of
coordinates.
Pseudo-Dilution: see Site
Isolation

